Vaginal Mesh: Class Actions, Trials and Tribulations
)
More than 6 percent of people in first world countries have received an implantable medical device "for rebuilding body functions, achieving a better quality of life, or expanding longevity" and 1.7 million patients have been injured worldwide from 2005 to 2014. Nevertheless the innovations and continual improvements in medical devices mean that a majority of patients have received great benefit from these implants.
Up to 15% of women recipients of vaginal mesh experienced vaginal bleeding, discharge and vaginal discomfort resulting from the protrusion of the mesh through the overlying vaginal skin. An Australian class action against a major surgical supplier by over 800 women suffering from complications of vaginal mesh repair has been underway in the Federal Court since 4 July 2017.Her Honour has yet to deliver her determination. The majority of the 800 patients in the class action are suffering severe vaginal/pelvic pain and many have experienced mesh erosion /exposure.
A critical question raised in the Trial was why the vaginal implantation of foreign material was introduced with so little prior assessment of its safety.
Vaginal mesh was introduced in the late 1990's, initially in thin bands 7 mm in width placed underneath the mid portion of the urethra to cure urinary incontinence. It proved to be highly successful in curing urodynamic stress incontinence with long term cures of up to 92%. That experience informed the TGA deliberations on vaginal prolapse mesh implantations.
In 2015 a Dutch journalist Jet Schouten in cooperation with Professor Carl Heneghan an Oxford University expert in evidence based medicine, submitted a scam proposal to the Austrian branch of TUVan EU assessor-to approve a vaginal mesh that is normally used to package mandarins for use in humans. It received a tacit approval from TUV. The TGA relies on international agencies for much of its device assessments.
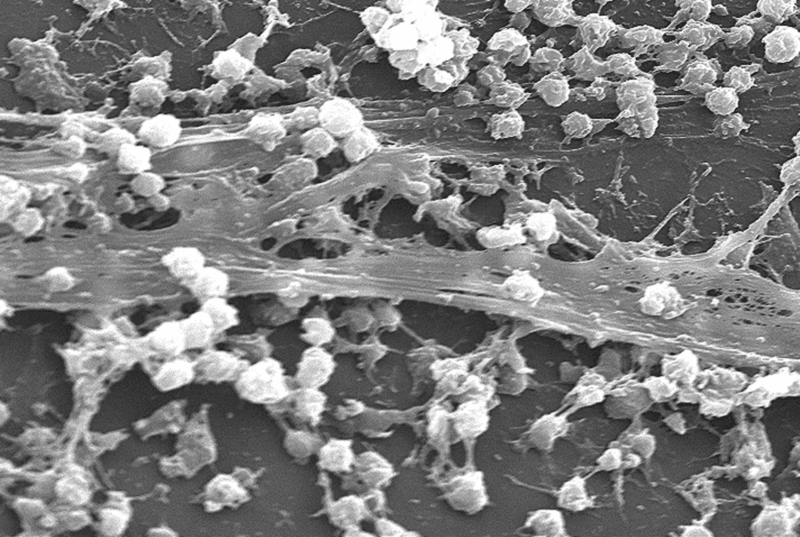
Klinge et al in 1999 evaluated a remarkable number of explanted meshes and found that the mesh porosity was the most important determinant of the tissue reaction and risk of scar entrapment. The pore size must be much larger than 75 microns to preserve tissue integration without filling the pores with scar tissue. Mesh contraction, which ultimately occurs in more than 25% of patients over time, is another cause for vaginal symptoms such as pain.
Biofilm: bacterial contamination of mesh
A biofilm refers to complex communities of bacteria residing within an exopolysaccharide matrix that adheres to a mesh surface. Colonisation of vaginally implanted mesh occurs frequently: in a study by Vollebregt et al in 2009, 83.6% of a series of vaginal mesh implants in 64 consecutive women were found to be contaminated with bacteria at the time of implantation. They were admittedly in low bacterial densities, irrespective of the sterility of the surgery.
There is a close correlation between chronic inflammation due to a foreign body and low grade chronic infection. Experience with other foreign body mesh insertions suggest that bacterial colonisation in the biofilm around implanted material may lead to low grade chronic infections. Host tissue erosion and mesh migration are very likely to be linked to contamination with microorganisms such as bacteria and fungi, with or without biofilm formation.
Characteristics of the Ideal Mesh
The ideal tissue substitute for prolapse repair is biocompatible, inert, inexpensive, induces minimal inflammatory response and, at the same time, acts as a scaffold to facilitate fibrous tissue ingrowth, is resistant to infection, avoids shrinkage and is easy to handle. The mesh should incur minimal inflammatory reaction and be followed by vascular and fibroblastic ingrowths. The key factors for host response to synthetic meshes are pore size and weave. The pore size must be much larger than 75 microns to preserve tissue integration without filling the pores with scar tissue. A pore size > 1 mm is required for prolapse, and the pore size should be > 3 mm in cases of mechanical strain. Meshes with large pores exhibit less inflammatory infiltrate, connective tissue, fistula formation, calcification, and bridging (i.e., the pores are filled by scar tissue) than meshes with small pores.
Abdominal Mesh Hernia Repairs: failure to appreciate mesh complications
The use of mesh for abdominal hernias especially inguinal hernias and incisional hernias commenced in 1958 when Usher et al published their technique using Marlex a material which because of its large pores, allowed granulation tissue to grow through it. Over time, fibroblast activity increased, more collagen was deposited without giant cells and the repair increased in strength over a 12 month period. Usher in 1963 reported the use of polypropylene mesh (a mono-filamentous polymer) for mesh abdominal herniorrhaphies. By 2003 it was apparent that mesh hernia repairs could cause significant chronic pain mediated by a foreign body reaction including inflammation, fibrosis, calcification, thrombosis or infection. The proportion of recipients who developed chronic pain was 'high' and typically presented more than 12 months after the herniorrhaphy. A syndrome of 'mesh inguinodynia' chronic pain at the site of mesh inguinal hernia repair was described in 1998. Narrow pore size and heavy fibre content were at that time implicated in the production of chronic pain. Light weight mesh products with large pore size (>1 mm) and elasticity of 25-35% at 16 Newtons per cm produced significantly reduced rates of chronic pain. This introduction of lightweight mesh for herniorrhaphies was motivated by a desire to 'restor[e] the abdominal functions and be integrated physiologically into the abdomen based on a maximum of biocompatibility'.
Australian response to vaginal mesh
In Australia the TGA decided on 28 November 2017 to remove transvaginal mesh products whose sole use is the treatment of pelvic organ prolapse by implantation from the Australian Register of Therapeutic Goods (ARTG). This effectively deprived gynaecologists of using vaginal mesh. Since the review commenced in 2013, 45 devices have been removed from uro-gynaecological use by the TGA - 43 cancelled from the ARTG and a further two have been limited to non-uro-gynaecological procedures. The TGA also responded to the concerns regarding the insertion of vaginal mesh by reclassifying such devices from Class II b (medium to high risk) to Class III (high risk) in December 2018.
End users (surgeons) have the obligation to be fully apprised of the characteristics of implanted products. This should guide the selection of mesh product and the selection of patients who could benefit from mesh implants. Surgeons also have the obligation to ensure that the surgical skills with which they implant mesh are conscientiously learnt and upgraded regularly. The designers, manufacturers and regulatory bodies for medical devices are reliant on surgeons for clinical research especially audits of mesh recipients' complications and efficacy. That information is critical for patient safety.
From a surgical perspective it seems likely that vaginal mesh will be abandoned in favour of laparoscopic abdominal colpopexies or vaginal sacrospinous fixations.
Further reading:
-
Johnson & Johnson / Ethicon Class Action
https://www.shine.com.au/service/class-actions/johnson-johnson-ethicon-class-action retrieved 19 July 2019 - Klinge U, Klosterhalfen B, Muller M, Schumpelick V. Foreign body reaction to meshes used for the repair of abdominal wall hernias. Eur J Surg. 1999;165:665673
-
Ford AA, Rogerson L, Cody JD, Aluko P, Ogah JA. Mid-urethral sling operations for stress urinary incontinence in women. Cochrane Database of Systematic Reviews 2017, Issue 7. Art. No.: CD006375. DOI: 10.1002/14651858.CD006375.pub4
https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD006375.pub4/epdf/full retrieved 21 July 2019 - Atherton M, Daborn J, Tsokos N, Jeffery JT, Yin M. Complications associated with tissue anchor migration after vaginal surgery using the tissue fixation system a case series. Australian and New Zealand Journal of Obstetrics and Gynaecology 2012; 52:83-86.
- Maher C, Feiner B, Baessler K, ChristmannSchmid C, Haya N, Marjoribanks J. Transvaginal mesh or grafts compared with native tissue repair for vaginal prolapse. Cochrane Database of Systematic Reviews 2016, Issue 2. Art. No.: CD012079. DOI: 10.1002/14651858.CD012079.
-
Scandal of fruit netting 'approved as surgical implant'(London, Sunday Times Jan 11.2015)
https://www.thetimes.co.uk/article/scandal-of-fruit-netting-approved-as-surgical-implant-dvcqd2rt9mr retrieved 25 August 2019
| Tags:Medicolegal |








